Renewable Surfactants
Surfactants are widely used in household detergents, cleaners, agrochemicals, emulsifiers, foaming agents and personal care. The global surfactant market is estimated at $30 billion and is expected to grow annually at the rate of 3%.
Currently, the production of anionic surfactants like linear alkyl benzene sulfonates (LAS), which constitute about 50% of the total surfactants market, rely on petrochemical-based constituents, such as benzene and long-chain hydrocarbons.
At CCEI, the design of new surfactants has focused on bio-renewable technologies for producing surfactant molecules with high biodegradability, strong detergency, good solubility and improved stability in hard water and a range of temperatures. Through the Friedel-Crafts acylation reaction, sugar derived furans can be linked to triglyceride-derived long chain fatty acids (e.g. lauric acid) in high yields (>90%) using trifluoroacetic anhydride or heterogeneous catalysts such as zeolites. These molecules are then subsequently hydrogenated to form alkylfurans that structurally resemble linear alkylbenzene molecules. Additionally, branching can be introduced in the long chain via aldol condensation with smaller aldehydes post acylation followed by hydrogenation to achieve desired surfactant properties. The hydrophilic group is then attached to the hydrophobic alkylfuran molecule (e.g. via sulfonation) to synthesize the oleo-furan surfactant.
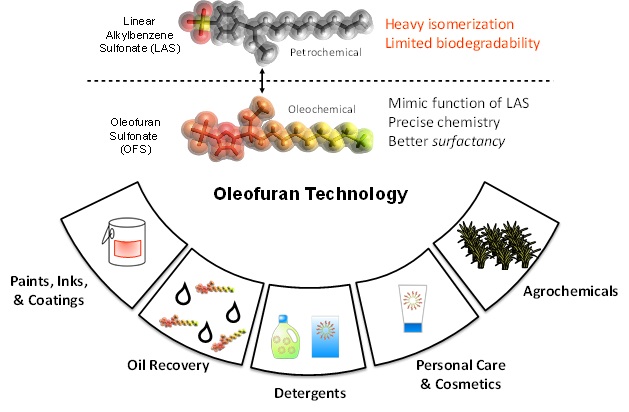
This technology differs from the existing process in that the acylated aromatic products do not easily isomerize or continue to acylate to form side products, whereas petroleum-derived alkylated benzene molecules tend to isomerize, forming products with alkyl branches with variable length, thereby reducing selectivity toward desired products. The high selectivity and tunability of the process enables precise synthesis of surfactants with predefined properties from bio-renewable feedstock. Besides, based on experimental results, alkylfuran based surfactants have been found to possess improved detergency at low concentrations and in cold water and drastically enhanced hard water stability. The limited selectivity and tunability of LAS surfactants resulting in poor hard water performance has demanded the use of expensive chelating agents in detergent formulations. Furan-based surfactants, on the other hand, have demonstrated up to an order of magnitude improvement in hard water stability than the conventional surfactants, thereby bypassing the need for chelants. Tunable functionalities of furan and varying lengths of fatty acids enable the synthesis of a wide variety of surfactants that find applications in myriad sectors.
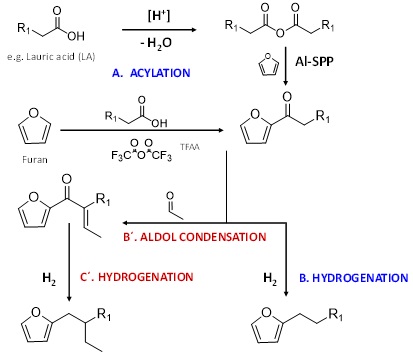
Figure: Reaction pathway for synthesis of oleo-furan surfactants
